Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (23): 3645-3652.doi: 10.3969/j.issn.2095-4344.2014.23.007
Previous Articles Next Articles
Treatment of spinal cord injury by transfection of double gene recombinant adenovirus vector into rat bone marrow mesecnhymal stem cells
Xu Yan-mei1, Sun Fei2, Hui Chun-ying3, Wang Wei4
- 1Department of Critical Care Medicine, 2Department of Emergency, 3Department of Cardiac and Brain Surgery, 4Department of Rehabilitation Medicine, First Affiliated Hospital of Liaoning Medical University, Jinzhou 121001, Liaoning Province, China
-
Revised:2014-03-08Online:2014-06-04Published:2014-06-04 -
Contact:Wang Wei, Master, Associate professor, Department of Rehabilitation Medicine, First Affiliated Hospital of Liaoning Medical University, Jinzhou 121001, Liaoning Province, China -
About author:Xu Yan-mei, Senior nurse, Department of Critical Care Medicine, First Affiliated Hospital of Liaoning Medical University, Jinzhou 121001, Liaoning Province, China -
Supported by:the grant from the Social Development Department of Liaoning Science and Technology Bureau, No. 2012408002
CLC Number:
Cite this article
Xu Yan-mei, Sun Fei, Hui Chun-ying, Wang Wei. Treatment of spinal cord injury by transfection of double gene recombinant adenovirus vector into rat bone marrow mesecnhymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(23): 3645-3652.
share this article

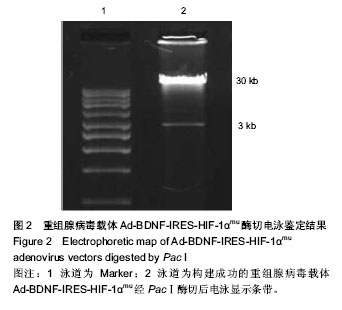
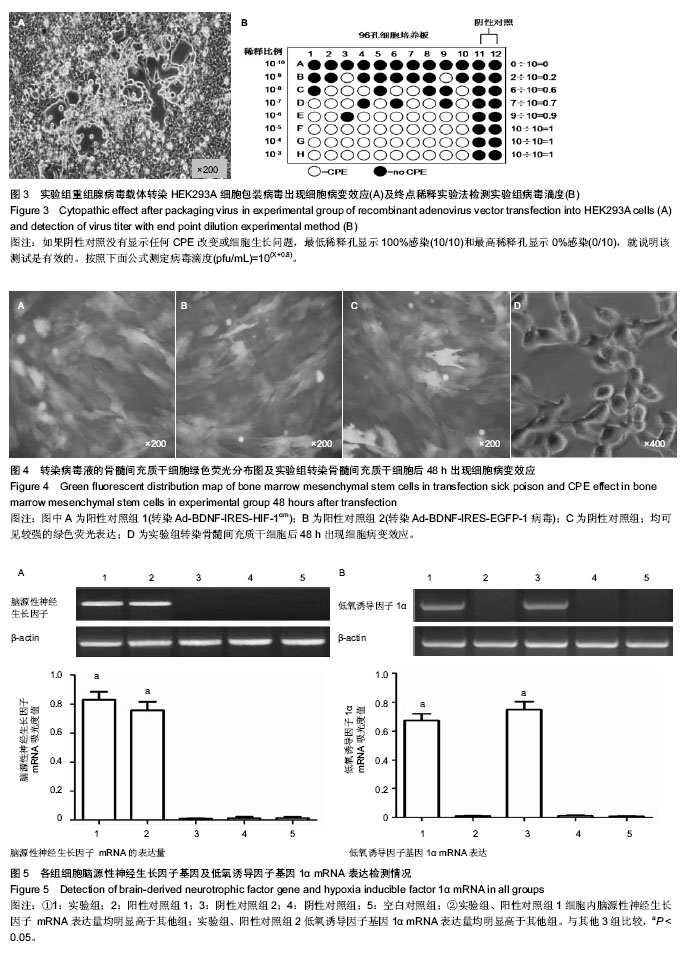
2.1 测序及腺病毒载体鉴定 基因测序显示低氧诱导因子1α基因编码区第402位脯氨酸由CCA突变成GCA(丙氨酸),第564位脯氨酸由CCC突变成GCC(丙氨酸),第803位天冬酰胺由ATT突变成AGC丙氨酸(说明:第803位天冬酰胺测序的是反义链,所以天冬酰胺正义链应为AAT,而丙氨酸正义链应为GCT)(图1)。凝胶电泳成像显示Ad-BDNF- IRES-HIF-1αmu经PacⅠ酶切后出现30 kb和3 kb两个条带,提示重组双基因单腺病毒载体构建成功(图2)。 2.2 腺病毒包装及滴度测定 重组病毒体经Lipofectamine 2000转染HEK293A细胞后均出现细胞病变效应(Cytopathic effect,CPE),具体体现在细胞触角回收、肿胀变圆,一部分细胞脱落,悬浮于视野中(图3A)。 包装成功的病毒液采用终点稀释实验法(End-Point Dilution Assay)进行病毒滴度测定(图3B),病毒滴度检测结果依次为实验组(1.6×108 pfu/mL),阳性对照组1(1.9× 108 pfu/mL),阳性对照组2(1.4×108 pfu/mL)和阴性对照组(2.0×108 pfu/mL),均符合后续转染实验要求。 2.3 病毒转染骨髓间充质干细胞表达结果 包装成功的两组阳性对照及一组阴性对照病毒液在荧光显微镜下均观察到较强绿色荧光表达,转染效率较高(图4A-C);根据对照组转染效果进行实验组病毒液的转染,48 h后可见大鼠骨髓间充质干细胞发生细胞病变效应,转染效率依然很高(图4D)。 2.4 RT-PCR结果 脑源性神经生长因子 mRNA表达:实验组、阳性对照组1相对A值分别为0.81±0.06,0.75±0.07,两组比较差异无显著性意义(P > 0.05);而另外3组脑源性神经生长因子mRNA的表达量非常低,几乎可以忽略(P > 0.05);实验组、阳性对照组1两组表达量均明显高于其他3组 (P < 0.05,图5A)。 低氧诱导因子1α mRNA表达:实验组、阳性对照组2表达量分别为0.69±0.05,0.73±0.08,两组之间比较差异无显著性意义(P > 0.05);而另外3组低氧诱导因子1α mRNA的表达量基本没有(P > 0.05);实验组、阳性对照组2两组表达量均明显高于其他3组(P < 0.05,图5B)。"

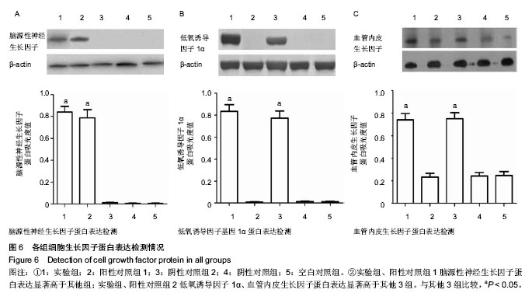
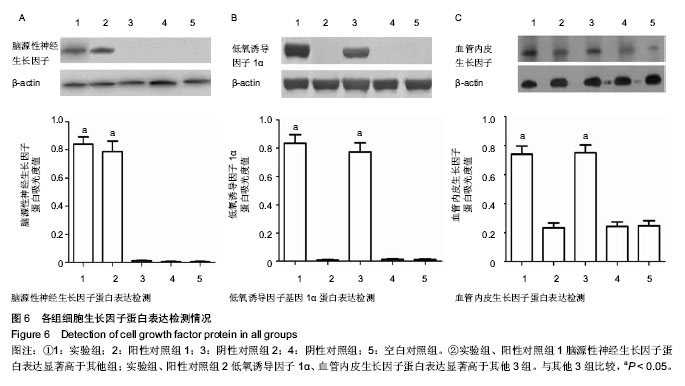
2.5 Western blot结果 脑源性神经生长因子蛋白表达:实验组、阳性对照组1两组蛋白表达相对A值分别为0.82±0.08和0.79±0.05;相对于其他3组均有显著性意义(P < 0.05);而阳性对照组2、阴性对照组、空白对照组3组蛋白表达量非常低,组间相比差异无显著性意义(P > 0.05),与mRNA检测结果相符(图6A)。 低氧诱导因子1α蛋白表达:实验组、阳性对照组2两组相对A值分别为0.83±0.05和0.77±0.08;相对于其他3组均有显著性意义(P < 0.05);而阳性对照组1、阴性对照组、空白对照组3组之间差异无显著性意义(P > 0.05),见图6B。 血管内皮生长因子蛋白表达:实验组、阳性对照组2两组相对A值分别为0.74±0.06和0.72±0.03;相对于另外三组均有显著性意义(P < 0.05);而阳性对照组、阴性对照组、空白对照组3组之间差异无显著性意义(P > 0.05),见图6C。 以上结果提示,三点突变型低氧诱导因子1α不仅可以在常氧条件下高效表达,还能够促进其下游基因血管内皮生长因子的高效表达。"
| [1] Grossman SD, Rosenberg LJ, Wrathall JR. Temporal-spatial pattern of acute neuronal and glial loss after spinal cord contusion. Exp Neurol. 2001;168(2):273-282. [2] Herbertson A, Aubin JE. Cell sorting enriches osteogenic populations in rat bone marrow stromal cell cultures. Bone. 1997;21(6):491-500. [3] Nishimura R, Okuda K. Hypoxia is important for establishing vascularization during corpus luteum formation in cattle. J Reprod Dev. 2010;56(1):110-116. [4] Fiorenzo P, Mongiardi MP, Dimitri D, et al. HIF1-positive and HIF1-negative glioblastoma cells compete in vitro but cooperate in tumor growth in vivo. Int J Oncol. 2010; 36(4):785-791. [5] DE Lima PO1, Jorge CC, Oliveira DT, et al. Hypoxic condition and prognosis in oral squamous cell carcinoma. Anticancer Res. 2014;34(2):605-612. [6] Liang YC, Wu HG, Xue HJ, et al. Effects of PI3K inhibitor NVP-BKM120 on acquired resistance to gefitinib of human lung adenocarcinoma H1975 cells. J Huazhong Univ Sci Technolog Med Sci. 2013;33(6):845-851. [7] Friedenstein AJ, Chailakhyan RK, Gerasimov UV. Bone marrow osteogenic stem cells: in vitro cultivation and transplantation in diffusion chambers. Cell Tissue Kinet. 1987; 20(3):263-272. [8] Zohar R, Sodek J, McCulloch CA. Characterization of stromal progenitor cells enriched by flow cytometry. Blood. 1997;90(9): 3471-3481. [9] Tropel P, Noël D, Platet N, et al. Isolation and characterisation of mesenchymal stem cells from adult mouse bone marrow. Exp Cell Res. 2004;295(2):395-406. [10] Wan C, Gilbert SR, Wang Y, et al. Activation of the hypoxia-inducible factor-1-alpha pathway accelerates. Proc Nat Acad Sci. 2008;105(2):686-691. [11] Giaccia A , Siim BG , Johnson RS. HIF-1 as a target for drug development. Nat Rev Drug Discov. 2003;2(10):803-811. [12] Bai L, Yang JC, Ok JH, et al. Simultaneous targeting of Src kinase and receptor tyrosine kinase results in synergistic inhibition of renal cell carcinoma proliferation and migration. Int J Cancer. 2012;130(11):2693-2702. [13] Konstantina A, Lazaris AC, Ioannidis E, et al. Immunohistochemical expression of VEGF, HIF1-a, and PlGF in malignant melanomas and dysplastic nevi. Melanoma Res. 2011;21(5):389-394. [14] Coma S, Shimizu A, Klagsbrun M. Hypoxia induces tumor and endothelial cell migration in a semaphorin 3F- and VEGF-dependent manner via transcriptional repression of their common receptor neuropilin 2. Cell Adh Migr. 2011;5(3): 266-275. [15] Bruick RK , McKnight SL . A conserved family of prolyl-4-hydroxylases that modify HIF. Science. 2001; 294 (5545):1337-1340. [16] Evans CE, Humphries J, Mattock K, et al. Hypoxia and upregulation of hypoxia- inducible factor 1{alpha} stimulate venous thrombus recanalization. Arterioscler Thromb Vasc Biol. 2010;30(12):2443-2451. [17] Lando D , Peet DJ , Whelan DA , et al. Asparagine hydroxylation of the HIF transactivation domain a hypoxic switch. Science. 2002;295(5556):858-861. [18] Liu X, Du J, Xi Q. HIF-1α ODD polypeptides increased the expression of HIF1 and VEGF in hypoxic rat cortical neuron. Neurol Sci. 2011;32(6):1029-1033. [19] Toussaint M, Fievez L, Desmet CJ, et al. Increased hypoxia-inducible factor 1α expression in lung cells of horses with recurrent airway obstruction. BMC Vet Res. 2012;8:64. [20] Friedenstein AJ, Gorskaja JF, Kulagina NN. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol. 1976;4(5):267-274. [21] Muñoz-Elías G, Woodbury D, Black IB. Marrow stromal cells, mitosis, and neuronal differentiation: stem cell and precursor functions. Stem Cells. 2003;21(4):437-448. [22] Dougherty KD, Dreyfus CF, Black IB. Brain-derived neurotrophic factor in astrocytes, oligodendrocytes, and microglia/macrophages after spinal cord injury. Neurobiol Dis. 2000;7(6 Pt B):574-585. [23] Pereira RF, Halford KW, O'Hara MD, et al. Cultured adherent cells from marrow can serve as long-lasting precursor cells for bone, cartilage, and lung in irradiated mice. Proc Natl Acad Sci U S A. 1995;92(11):4857-4861. [24] Emans PJ , Spaapen F , Surtel DA , et al. A novel in vivo model to study endochondral bone formation;HIF-1α activation and BMP expression. Bone. 2007;40(2):409-418. [25] Huang SM, Chen TS, Chiu CM, et al. GDNF increases cell motility in human colon cancer through VEGF-VEGFR1 interaction. Endocr Relat Cancer. 2013;21(1):73-84. [26] Zimmermann SM, Würgler-Hauri CC, Wanner GA, et al. Echinomycin in the prevention of heterotopic ossification - an experimental antibiotic agent shows promising results in a murine model. Injury. 2013;44(4):570-575. [27] Rodrigues B, Jorge L, Mostarda CT, et al. Aerobic exercise training delays cardiac dysfunction and improves autonomic control of circulation in diabetic rats undergoing myocardial infarction. J Card Fail. 2012;18(9):734-744. [28] Medina Villaamil V, Aparicio Gallego G, Santamarina Caínzos I, et al. Searching for Hif1-α interacting proteins in renal cell carcinoma. Clin Transl Oncol. 2012;14(9):698-708. [29] Huang C, Hales BF. Effects of exposure to a DNA damaging agent on the hypoxia inducible factors in organogenesis stage mouse limbs. PLoS One. 2012,7(12):e51937. [30] Weijts BG, Bakker WJ, Cornelissen PW, et al. E2F7 and E2F8 promote angiogenesis through transcriptional activation of VEGFA in cooperation with HIF1. EMBO J. 2012;31(19): 3871-3884. [31] Lin J, Qin X, Zhu Z, et al. FHL family members suppress vascular endothelial growth factor expression through blockade of dimerization ofHIF1α and HIF1β. IUBMB Life. 2012;64(11):921-930. [32] Lim W, Cho J, Kwon HY, et al. Hypoxia-inducible factor 1 alpha activates and is inhibited by unoccupied estrogen receptor beta. FEBS Lett. 2009;583(8):1314-1318. [33] Wu WJ, Zhang XK, Zheng XF, et al. SHH-dependent knockout of HIF-1 alpha accelerates the degenerative process in mouse intervertebral disc. Int J Immunopathol Pharmacol. 2013;26(3):601-609. [34] Wang X, Wang G, Kunte M, et al. Modulation of angiogenesis by genetic manipulation of ATF4 in a mouse modulation of angiogenesis by genetic manipulation. Invest Ophthalmol Vis Sci. 2013;54(9):5995-6002. [35] Artini PG, Simi G, Ruggiero M, et al. DHEA supplementation improves follicular microenviroment in poor responder patients. Gynecol Endocrinol. 2012;28(9):669-673. [36] Larrieu-Lahargue F, Welm AL, Bouchecareilh M, et al. Blocking Fibroblast Growth Factor receptor signaling inhibits tumor growth, lymphangiogenesis, and metastasis. PLoS One. 2012;7(6):e39540. [37] Tao X, Zhao N, Jin H, et al. FSH enhances the proliferation of ovarian cancer cells by activating transient receptor potential channel C3. Endocr Relat Cancer. 2013;20(3):415-429. [38] Shan B, Schaaf C, Schmidt A, et al. Curcumin suppresses HIF1A synthesis and VEGFA release in pituitary adenomas. J Endocrinol. 2012;214(3):389-398. [39] Zhang Z, Wang G, Li C, et al. Construction and characterization of a recombinant human adenovirus vector expressing bone morphogenetic protein 2. Exp Ther Med. 2013;6(2):329-334. [40] Romano G , Michell P , Pacilio C, et al. Latest developments in gene transfer technology:achievements,perspectives,and controversies over therapeutic applicat- ions. Stem Cells. 2000; 18(1):19-39. [41] Seregin SS, Amalfitano A . Overcoming pre-existing adenovirus immunity by genetic engineering of adenovirus-based vectors. Expert Opin Biol Ther. 2009; 9(12):1521-1531. [42] Douglas JT . Adenoviral vectors for gene therapy. Mol Biotechnol. 2007;36(1):71-80. [43] Elson DA , Thurston G , Huang LE , et al. Induction of hypervascularity without leakage or inflammation in transgenic mice overexpressing hypoxia-inducible factor-1alpha. Genes Dev. 2001;15(19):2520-2532. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [5] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [6] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [7] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [8] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [9] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [10] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [11] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [12] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [13] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [14] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [15] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||